Russia makes first batch of vaccine
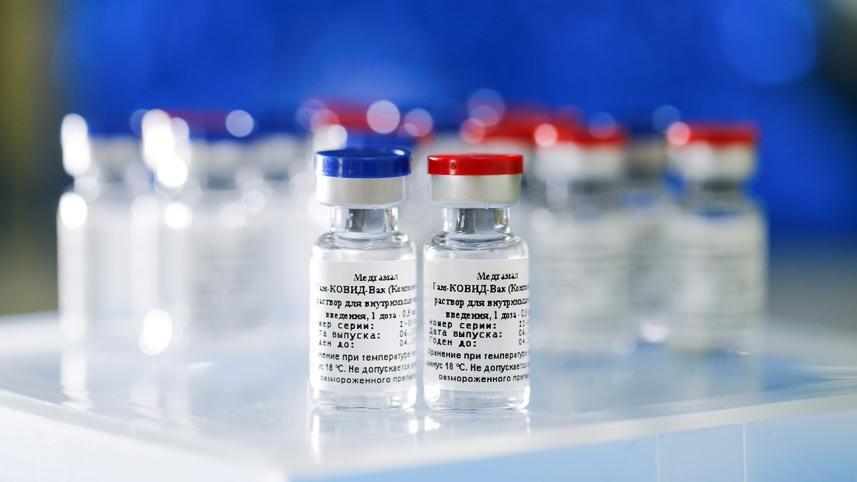
Russia has produced the first batch of its new vaccine for Covid-19, the Interfax news agency quoted the health ministry as saying yesterday, hours after the ministry reported the start of manufacturing.
President Vladimir Putin on Tuesday announced it had been first in the world to approve a vaccine. But the announcement was met with caution from scientists and the World Health Organization who said it still needed a rigorous safety review.
"The first batch of the novel coronavirus vaccine developed by the Gamaleya research institute has been produced," the health ministry said in a statement quoted by Russian news agencies.
Putin said the vaccine was safe and that one of his own daughters had been inoculated, though clinical trials were not yet complete and final stage testing involving more than 2,000 people only started this week.
Western scientists were sceptical, with some warning that moving too quickly on a vaccine could be dangerous, but Russia denounced criticism as an attempt to undermine Moscow's research.
The Russian vaccine is called "Sputnik V" after the Soviet-era satellite that was the first launched into space in 1957.
It was developed by the Gamaleya research institute for epidemiology and microbiology in Moscow in coordination with the Russian defence ministry.
The head of the institute, Alexander Gintsburg, told the TASS state news agency yesterday that volunteers taking part in the final stage testing of the vaccine's safety and efficacy would have two inoculations.
Russia has said that industrial production is expected from September and that it plans to manufacture 5 million doses per month by December or January.
Health Minister Mikhail Murashko said this week that the vaccine would first be made available to medics and would later be available to all Russians on a voluntary basis.
With more than 917,000 confirmed infections, Russia's coronavirus caseload is currently fourth in the world after the United States, Brazil and India.
Currently Russia has 92,000 people hospitalised with the virus and 2,900 in intensive care, according to the health ministry.
'CHALLENGE' TRIALS
US scientists are developing a strain of the coronavirus that could be used to deliberately infect volunteers in so-called "challenge studies," a government agency said Friday.
The work is preliminary and the government is continuing to prioritise randomised clinical trials of vaccine candidates, the National Institute for Allergies and Infectious Diseases (NIAID) said.
Several of these have entered their final stages, including the vaccines developed by Moderna, Pfizer and AstraZeneca.
But NIAID has nonetheless "begun efforts to manufacture a strain that could be used to develop a human challenge model, if needed," it said in a statement.
In normal clinical trials, volunteers receive either a medicine or a placebo and their health is then followed over the course of months of years.
Scientists look for how well the vaccine or treatment worked when the person was naturally exposed to the pathogen.
A quicker way to test whether a drug works is by deliberately infecting volunteers, as has been done in the past for influenza, malaria, typhoid, dengue fever, and cholera.
In the US, advocacy groups like 1DaySooner support challenge studies for Covid-19, but the subject is controversial because of how serious the disease can be and because its effects aren't fully understood.
NIAID said it would probably reach a decision toward the end of 2020, when the late-stage clinical trials that are underway start reporting their results.
This would help it determine whether challenge studies are needed, safe and ethical, it added.
David Diemert, the director of George Washington University's vaccine trial research unit who is overseeing a trial of Moderna's vaccine in the US capital, told AFP he did not think challenge trials were appropriate for Covid-19.
"I think they're a critical tool, but only under the right circumstances," he said.
Diemert is himself leading a challenge trial into a hookworm vaccine.
But he said that for Covid-19, "we don't have a very clear understanding of who is at risk of developing severe disease and we have no treatment that is guaranteed to cure someone if they do develop severe disease."
There's also enough widespread community transmission in the US -- the worst hit country in the world with 5.3 million confirmed cases -- to mean challenge trials are not necessary, he added.


 For all latest news, follow The Daily Star's Google News channel.
For all latest news, follow The Daily Star's Google News channel.
Comments