India’s Serum Institute to halt phase-3 Covid-19 vaccine trials
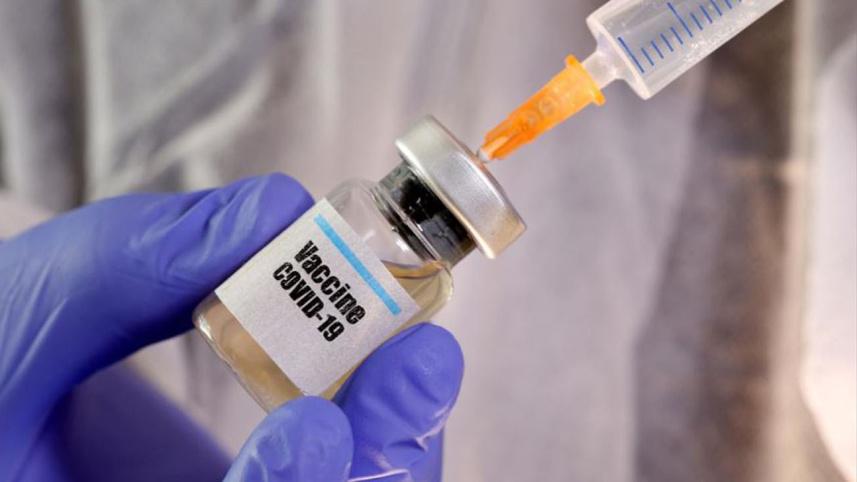
Serum Institute of India, which is producing and conducting trials of the Covid-19 vaccine being developed by Oxford University along with pharma giant AstraZeneca, today announced it will halt the Phase 3 trials set to begin in India next week.
"We are reviewing the situation and pausing India trials till AstraZeneca restarts the trials," read a statement from Pune-based Serum Institute of India which was doing the groundwork for the trial including listing of volunteers.
The trials were to be held on 1,600 volunteers across 17 sites in India and were expected to begin next week.
Bangladesh's Beximco Pharmaceuticals had recently struck a deal with Serum Institute of India (SII) to ensure the supply of Covid-19 vaccine being developed by the Indian drug manufacturer.
The Oxford vaccine candidate trial was stopped across four countries as a precautionary measure after one of the recipients in UK showed some potentially adverse symptoms.
AstraZeneca, the British-Swedish pharma giant with which SII has partnered with, had said the volunteer had suffered "an unexplained illness" and such pauses in drug trials are "routine".
The Serum Institute was issued a show-cause notice by India's drug sector controller DCGI which questioned why SII was continuing the trials in India even though it has been stopped elsewhere. The DCGI also questioned why it has not received a report detailing the symptoms of the patient in the UK.
"We are following DCGI's instructions and will not be able to comment further on trials. You can connect with DCGI for more updates on this front," said the SII statement signed by the institute headed by Adar Poonawalla.


 For all latest news, follow The Daily Star's Google News channel.
For all latest news, follow The Daily Star's Google News channel.
Comments